Precision and control at the level
of pharmaceutical requirements
Ensure quality control in the pharmaceutical industry with traceability, process validation, and data integrity to respond to audits.
Challenges in the pharmaceutical industry
The pharmaceutical industry operates in one of the most demanding environments in terms of quality, traceability, and validation. Every stage of the process must be rigorously controlled and documented, ensuring regulatory compliance and product safety. Among the main challenges are:
- Extreme traceability requirements for batches, samples, and operators
- Need for high-precision measurements and sampling
- Auditorias rigorosas e frequentes (GMP, ICH, FDA, EMA)
- Complex laboratory processes prone to human error
- High energy consumption in HVAC, controlled environments, and critical equipment

How ACCEPT addresses the sector’s challenges
ACCEPT provides an integrated ecosystem that ensures rigour, traceability, and control in highly regulated pharmaceutical environments.
-
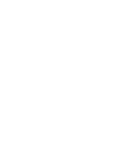
Full traceability of batches, samples, operators, and environmental conditions.
-
SPC to ensure process stability and detect micro-deviations before they impact a batch.
-
Validation of the accuracy and repeatability of measuring instruments.
-
Strengthening of CAPA systems with fully auditable documentation.
-
Monitoring of HVAC, sterilisation, compression, and refrigeration consumption.
-
Integrated view of quality, laboratory, and energy areas.
-
Integration with analytical equipment and automatic data acquisition, whenever applicable.
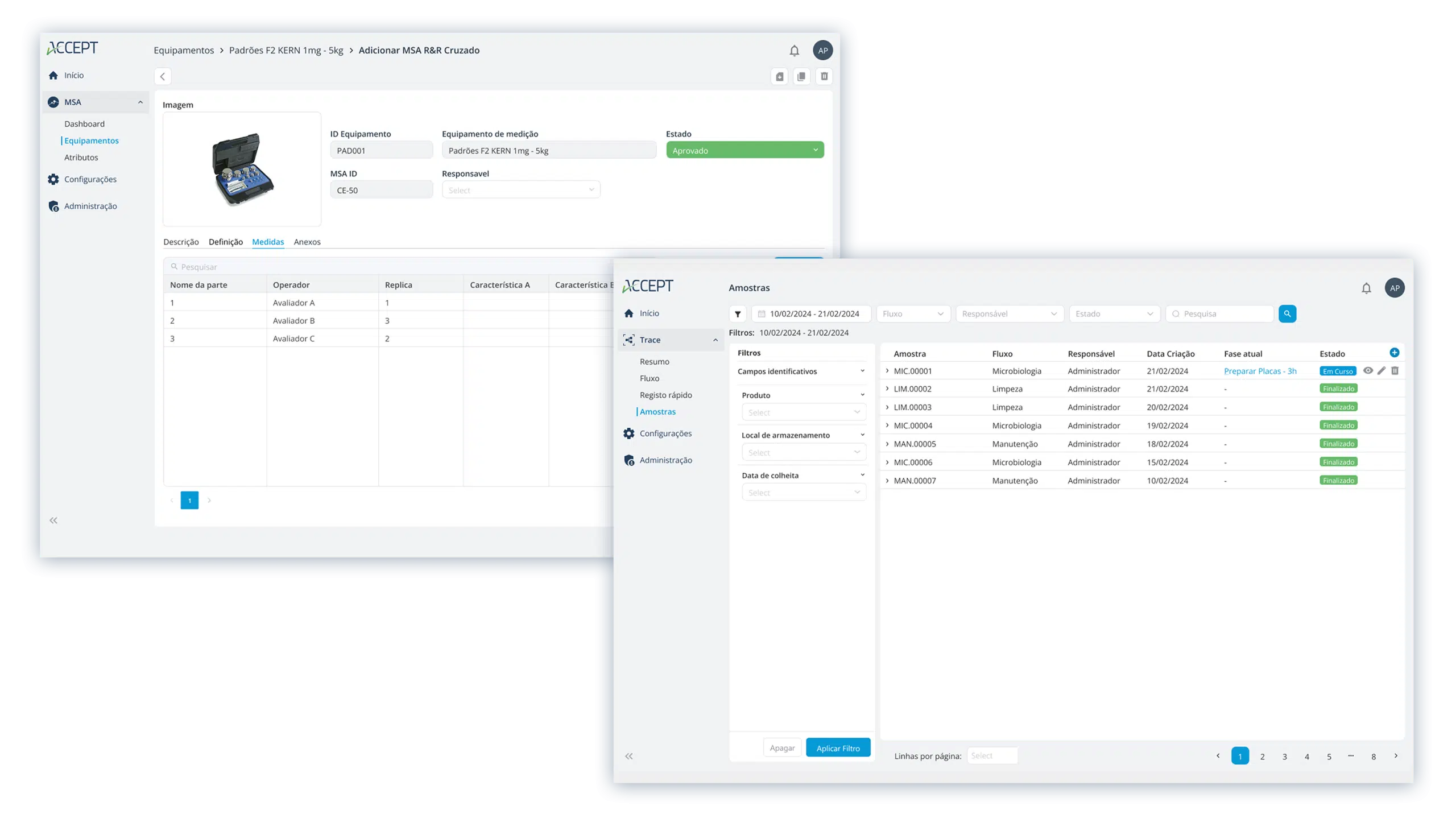
Essential applications for the pharmaceutical industry
All ACCEPT applications strengthen compliance, safety, and process consistency.
Direct benefits for the pharmaceutical industry
- Enhanced compliance with GMP, ICH, FDA, and regulatory requirements
- Reduced risk of critical deviations and batch impact
- Greater consistency and predictability across production runs
- Audit documentation always organised and readily available
- Reduction of energy costs in critical systems
- Integrated platform that reduces errors and increases process safety
Who is already using ACCEPT in the pharmaceutical industry




FAQs on Quality Control in the Pharmaceutical Industryacêutica
What is quality control in the pharmaceutical industry?
Quality control in the pharmaceutical industry involves ensuring that all products meet strict requirements for safety, efficacy, and regulatory compliance. It includes process monitoring, laboratory testing, and data validation throughout the entire production cycle.
What are Good Manufacturing Practices (GMP)?
Good Manufacturing Practices (GMP) are a set of standards that ensure pharmaceutical products are produced and controlled in a consistent and safe manner. These standards cover processes, documentation, equipment, and training, and are essential for ensuring quality and compliance.
How do you ensure GMP compliance in the pharmaceutical industry?
Ensuring GMP compliance involves controlling processes, ensuring complete traceability, and maintaining rigorous, auditable records. Continuous monitoring and data validation are essential for meeting regulatory requirements and responding to audits.
What is data integrity (ALCOA) in the pharmaceutical industry?
Data integrity refers to the guarantee that information is attributable, legible, contemporaneous, original, and accurate (ALCOA). This concept is essential for ensuring data reliability and meeting regulatory requirements in the pharmaceutical industry.
What is pharmaceutical process validation?
Pharmaceutical process validation consists of documented evidence that a process consistently produces results within defined criteria. It is an essential requirement for ensuring quality, safety, and regulatory compliance.
How do you ensure traceability in the pharmaceutical industry?
Traceability in the pharmaceutical industry allows the complete production history of a batch to be tracked, from raw materials through to the final product. This facilitates audits, ensures compliance, and allows the origin of any deviation to be quickly identified.
How do you improve quality control in the pharmaceutical industry with a digital solution?
Using a digital solution allows data to be centralized, traceability to be ensured, and information integrity to be maintained throughout the entire production process. Solutions like ACCEPT help strengthen quality control in the pharmaceutical industry, making audits easier and ensuring compliance with regulatory requirements.
Do you want to take your operation to the highest level of rigour?
Schedule a demo and see how ACCEPT supports compliance, validation, and efficiency in the pharmaceutical industry.